For safety managers, it is important to understand the dangers presented by chemicals before a worker begins handling them.
However, it can be challenging to ensure that everyone is protected all of the time – particularly when, given the nature of many hazardous chemicals, the problems created by risk tolerance are very real and workers may take the view that they can worry about the long-term effects of exposure later.
A holistic approach to chemical suit selection, maintenance and use is important in ensuring that protection is maintained when it should be.
This blog focuses on the critical information relating to chemicals that is vital for safety managers to understand before embarking on any task that could result in contamination.
The Importance of the Risk Assessment
Even before selection of a chemical suit the risk assessment provides the important first insight into the need for protection. It should be founded on the principle that the first action is to remove the hazard or risk entirely if possible (by replacing a chemical with a less hazardous option for example, or changing processes so the chemical is not required), or, failing that, to remove or minimize the risk of contamination occurring. Use of PPE should be the last line of defense.
It is important to understand the difference between risk and hazard (“risk” being the probability that a harmful event, such as contamination by a chemical will occur, and “hazard” being the consequences of that contamination if it does occur. It is in this last requirement – understanding the consequences of that contamination – where understanding the nature of the chemical itself become vital.
The subjects to consider in relation to the chemical fall into two categories:-
1. Understanding the properties of the chemical
2. Understanding how a chemical might penetrate the defense offered by chemical safety clothing, resulting in contamination of the wearer
Part 1: The Properties of the Chemical
There are perhaps over 8000 chemicals in use globally on a daily basis, each having a different and sometimes unique combination of properties. These properties can have an important influence both on how critical protection is and on how you can go about ensuring workers are protected. These properties can be summarized under the following headings:
- What is the hazard?
- Are the consequences long or short term?
- Will contamination be noticed at all?
- The chemical phase
- The chemical boiling point
- Likely routes of contamination
- What is the The Chemical Toxicity: Understanding Safe-Wear Times
Each of these subjects is discussed below.
What is the hazard and are the consequences long or short term?
The starting point is to understand what the hazard is; in other words, what are the consequences if a worker is contaminated and how long will they take to emerge?
The challenge with many chemicals – and what makes them different to most other workplace hazards – is that whilst some will have immediate, noticeable effects (acids may burn for example) many will cause long term health defects that are often life changing, not uncommonly life-ending and generally irreversible. Cancers, adverse effects on fertility, damage to internal organs and so on are consequences of many chemicals that may take years to take effect – long after it is far too late to ask if worker protection is good enough.
Understanding the consequences of contamination will guide users in several ways:-
- How critical is the protection? Clearly protecting against contamination by a chemical such as that in the “Dark Waters” movie is far more important than protecting against one that might have nothing more than minor short term effects such as skin irritation. This will also help to indicate how far protection can be compromised in favor of safety.
- How great is the danger of “risk tolerance”? This is the tendency of workers to develop an “it’ll be OK” attitude over time and consequently and take less care on a daily basis – clearly a much greater problem with chemicals that have only long term consequences. Dealing with this means greater emphasis on how garment use is managed using training and behavioral psychology.
| The America Petroleum Institute first declared that “the only safe exposure level for benzene is zero” in 1948. Yet it was the 1970s before the first regulations controlling benzene use emerged. |
It is also worth bearing in mind that understanding of the hazards of chemicals is commonly limited or even unrecognized. Too often catastrophic long term consequences are discovered after the event – and even then it may take years or even decades for studies to confirm any link (again, “Dark Waters” is a good example of this) and for controlling regulation to catch up. A review of safety data sheets of chemicals will often confirm this with the use of phrases such as “suspected of causing cancer” or “may damage the unborn child”. Basically, when comes to chemical hazards the jury is often out – or even not yet convened!
For this reason the wise safety manager will recognize that even if health consequences are not recognized now, they may well emerge later and so will build in wide safety margins when it comes to chemical protection – especially when considering safe-wear times (see below).
Will contamination be noticed at all?
What makes many chemicals doubly dangerous is that not only might contamination have only long term effects, but that should workers actually be contaminated, that is, if the chemical permeates or penetrates inside the garment and comes into contact with their skin… they may not notice at all. And if they happen to be doing a job that is repeated daily or weekly, they may be blissfully unaware of the regular contact, remaining so until the health problems develop months, years, or even decades later.
When chemicals feature this dangerous combination, the need to ensure that protection is effective – along with the need for managing and monitoring garment use through training and other methods – becomes much more important.
Chemical Phase: Liquid, Dust or Gas?
Whether the chemical is in liquid, dust, or gaseous form is important in determining the type of chemical protection required:-
- Liquid chemicals will require garments certified to CE Types 3, 4 or 6 depending on the type of liquid contact.
- Dry particle or dust chemicals may require a CE Type 5 garment. However, note that particulates are unlikely to permeate through chemical suit materials and will only enter the suit through a process of penetration – through holes or gaps in a fabric, the garment construction (such as the zip) or through gaps between the safety clothing and other items of PPE (between the suit and mask, the sleeves and gloves and so on).
In the case of dusts, because they float in the atmosphere and move with air flows, behaving differently to liquids, the issue of “the Bellows Effect”, can be critical.
It is also worth noting that dusts are more likely to present respiratory hazards than skin contact hazards. This does not mean however that the body protection provided by safety clothing is not required or important. In this case, a chemical suit can prevent dust contaminating the wearers own clothing or body only to be breathed in later. Secondary dust inhalation, not only by the wearer but also by the wearers family and friends, is now a well recognized problem.
Gases or vapors are likely to require a Type 1 suit – a fully encapsulated suit that is sealed against the environment so that gasses cannot penetrate.
However, note that choice of garment Type according to CE standards is always subject to the nature of a chemical – especially its toxicity. Protection against a highly hazardous chemical where contact is defined as Type 6, may require a much higher level of protection than a Type 6 suit – even to the extent of a gas-tight suit – as even the smallest level of contamination could be a problem.
The Boiling Point
The phase of any material is not fixed. Solids become liquids and liquids become gases at the right temperature. Water is solid ice at less than 0oC and gas at more than 100oC. The boiling point of chemicals can vary enormously. Ammonia notoriously boils, becoming gas, at a temperature (-35.5C) well below that of the freezing point of water and normal ambient temperatures. Liquids can also vaporize gradually as temperature rises depending on other conditions.
Thus the tendency of and temperature at which a liquid chemical will vaporize is an important consideration.
If a liquid chemical is identified as likely to become a vapor or gas at low temperature a standard liquid protective suit of Type 3,4 or 6 might not be enough and a gas tight or encapsulating suit might be appropriate.
Likely Routes of Contamination
How will the chemical enter the body?
Some chemicals can only enter the body via inhalation or ingestion. Many however will readily permeate through the skin and into the blood stream. Understanding how the chemical enters the body is another important factor in assessing the type of protection required.
However, again, just because the primary route may be through the mouth, nose or eyes does not mean that the body protection offered by a safety coverall is not needed or not important. The possibility of secondary contamination is always a risk, especially with hazardous dusts, so it can be important to ensure a chemical does not contaminate the wearers own clothes or skin.
Chemical Toxicity: How much chemical is required to cause harm?
The “Toxicity” of a chemical can be described as the amount of it that might result in harm should someone be contaminated with it.
Some chemicals are more dangerous than others. In some cases, it might be possible to come into contact with large volumes and suffer very little harm.
Other chemicals can cause a great deal of harm in only tiny volumes. Chemical Warfare Agents are deliberately synthesized to do just that and have the potential to kill or seriously incapacitate thousands of people with only very small volumes. Whilst no commercial chemicals are of this dramatic level of toxicity, many can have harmful consequences in relatively small amounts. Thus understanding how much of a chemical is required to cause harm, and therefor how much contact will NOT cause harm, is one of the most critical questions in chemical suit management.
|
Why is toxicity measured as a volume per surface area of skin? |
|
Why not simply measure a fixed volume? |
How is Toxicity Measured?
The toxicity of chemicals, in the case of dermal toxicity is measured in terms of the volume of contact in micrograms(µg) per cm2 of surface area of skin. This is not however an exact science for a variety of reasons (not least that different metabolisms mean toxicity limits may vary depending on the individual). However EU Regulation 1272: 2008 identifies many chemicals as having one of three general toxicity limits shown in the table below:-
| 20µg / cm2 | Highest level of Toxicity |
| 75µg / cm2 | Medium level of Toxicity |
| 150µg / cm2 | Lowest level of Toxicity |
These levels of toxicity allow users of chemical suits to make two types of decisions:-
1. General judgements about the hazard presented by the chemical and relative need to ensure protection – clearly a chemical with the highest toxicity level where only 20µg /cm2 may cause harm demands that ensuring protection is effectively maintained at all times is given a high priority.
2. It allows detailed judgements about how long wearers can safely work with a chemical before a harmful volume of contamination might be reached. Combined with a knowledge of the rate of permeation of a chemical through the fabric it allows calculation of effective safe-wear times. This is dealt with in the Second Part of this Blog below.
In summary, understanding the specific properties of the chemical as listed above contributes to assessment of the importance of protection, the type of chemical safety clothing required and the extent to which and how maintenance and use should be managed. However, the second question is to understand how the chemical might enter the suit and contaminate the wearer.
Part 2: How Might the Chemical Penetrate the Defenses of the Suit?
There are essentially two ways that this might happen: Penetration and Permeation.
Penetration and Permeation: What’s the Difference?
Whilst the net result is the same: the chemical gets inside the suit and contaminates the wearer, these process are different and understanding protection requires an understanding of that difference:-
Penetration: a macro-level process where the chemical passes through holes or gaps in the structure of the fabric, the garment construction (such as seams or zip) or gaps between the coverall and other PPE such as gloves and face-mask. Penetration deals with larger volumes and is more critical for chemicals that will cause harm in greater volumes or chemicals that will have immediate effects such as causing burns
Permeation: a micro-level process where a chemical passes through the apparently solid barrier of the fabric. The molecules of the chemical pass between those of the fabric material by a process of absorption and desorption. Permeation deals with very small volumes (micrograms) and is important for those chemicals harmful in small volumes.
The video below summarizes the difference:-
Whilst those selecting chemical suits often focus on the permeation of a chemical through a fabric it is important to realize that the much greater risk is that a chemical will penetrate through weaknesses in the construction of the garments itself or in the entire ensemble of PPE.
Penetration is the greater risk; safety clothing fabric that resists permeation will be useless if the possibility of penetration through weak construction is ignored or because of a failure to understand how effectively different types of PPE work together.
Understanding Permeation Rates and Safe-Wear Times
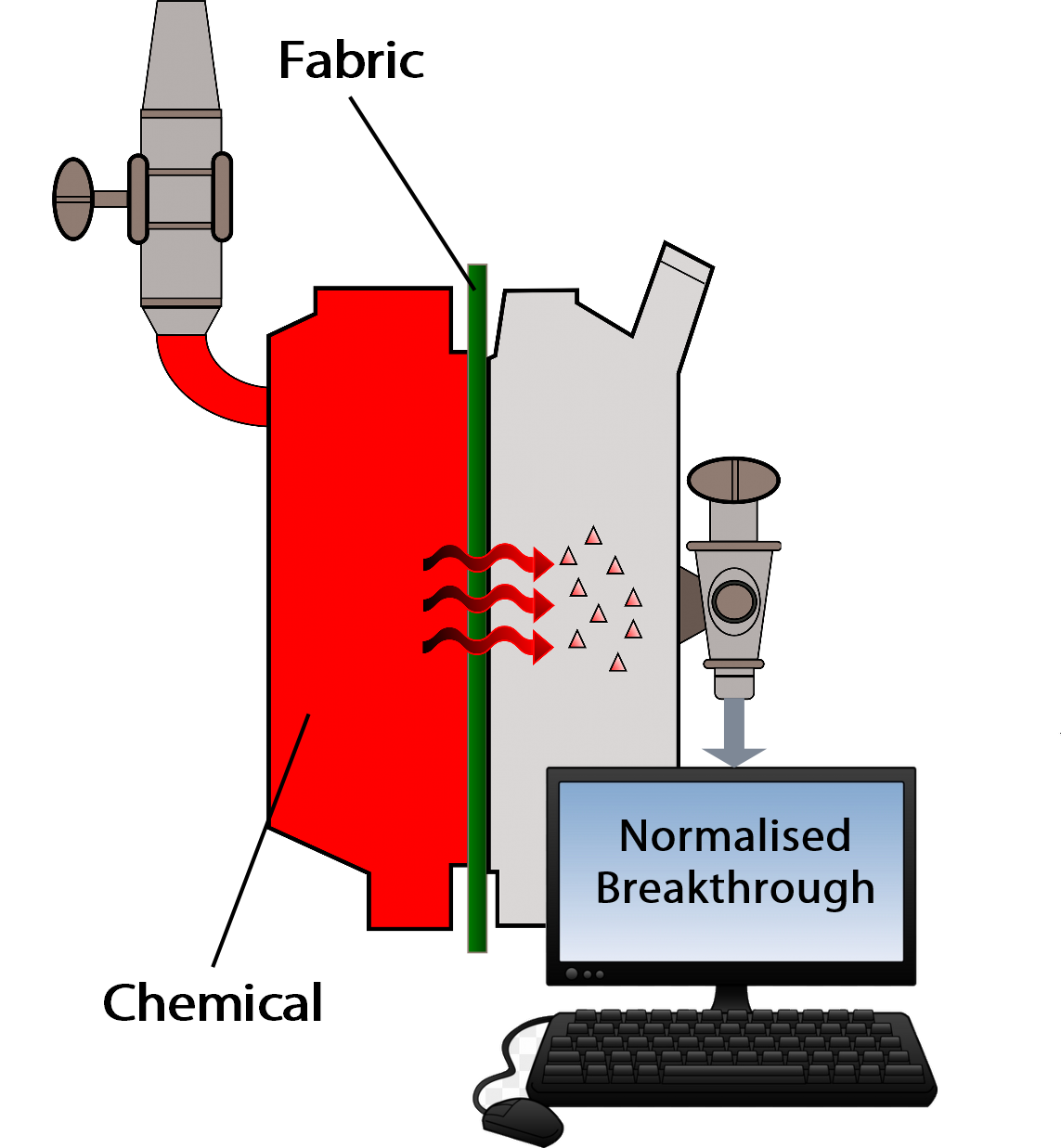
Possibly the biggest misunderstanding in PPE is the misinterpretation of the chemical permeation test (EN 6529) used to measure the ability of chemical suit fabric to resist permeation by a named chemical. Many selectors of chemical suits focus on the results of this test in the belief it tells them unequivocally that their workers are safe. In fact it does nothing of the sort.
| Permeation Test EN 6529 | |
|
|
The permeation test measures the rate of permeation of chemical through a suit fabric and the “Normalized Breakthrough” is the time until that rate reaches 1.0µg/cm2/minute. This does neither indicates when the chemical first breaks through the fabric nor a safe-wear time. Normalized breakthrough is only intended to allow performance comparison of different fabrics – not to indicate how long a suit can be safely worn, or even that a suit is safe to use. |
The belief is that the “breakthrough” result of a permeation test, for example a result of 360 minutes, suggests that no chemical has permeated through the fabric in that time so the suit can safely be used for up to 360 minutes. This is a misunderstanding; in fact the test measures a time until a RATE OF PERMEATION through the fabric is achieved and not when it first “breaks through” the fabric. This means that the chemical is permeating through before – possibly long before – that rate of penetration is reached. This might be an important fact for chemicals with a high toxicity.
How to Assess Safe-Wear Times
Thus, given that the permeation test does not indicate a safe wear time, how do users know how long a suit can be worn safely?
It is important to recognize that permeation of a chemical through an apparently “solid” barrier such as a chemical suit fabric will always happen; it cannot be prevented, only delayed and slowed. It is just a question of when and how quickly it occurs.
Given that the permeation test measures the RATE or SPEED of permeation through the fabric it can also be used to determine the volume of the chemical permeating through over time. This volume can be compared with the toxicity level of the chemical to assess how long it will take until a volume equal to its toxicity limit is reached.
In other words, this simple calculation provides users with a realistic safe-wear time; the maximum amount of time that a user can work with a chemical before a possibly dangerous level of permeated volume is reached.
| EN 14325 is a support standard that provides test references, methods and classification tables for all testing of chemical protective clothing. You can read more about this and different types of standards in our blog here. |
In fact, the new version of EN 14325 published in 2018 recognized this and provided a new system of classification of chemicals using the volume permeated and the chemical dermal toxicity to provide a new system of classification of chemical suits.
However, one point to bear in mind is that the rate of permeation through fabric will increase as temperature increases. A general rule of thumb is that permeation rate may double for every 10oC rise in temperature. This means that any calculation of safe-wear time may be incorrect if temperature in the environment varies substantially and that more chemical may be permeating than indicated. Users should therefor take this into account in any safe-wear time calculations.
You can read about how to calculate safe-wear times in our blog here.
Tools such as Permasure®, an app to calculate safe-wear times and outlined below, can be a useful addition to a safety managers’ range of weapons to help manage chemical suits.
The important take-away here is that the Normalized Breakthrough currently provided by chemical safety clothing manufacturers is NOT an indication of safety or safe-wear times but only a method of comparing performance of different suit fabrics. Especially where chemicals feature high toxicity, it is important to gain knowledge of permeation rates to assess real world safe-wear times and ensure users never wear suits to handle chemicals for longer than the time it may take to reach dangerous levels of contamination through permeation. (And remember, given the nature of many chemicals, that contamination may not even be noticed!)
CONCLUSION: A Holistic Approach
The common focus on permeation test results of those involved in selecting and managing chemical safety clothing is important but cannot be taken in isolation as an indication that a suit will provide the protection needed.
Safety managers need to understand the limitations of permeation test results and go further, considering the issues of chemical toxicity and realistic safe-wear times, whilst developing a good understanding of the nature and properties of – and especially the hazard presented by – any on-site chemicals that workers need protection from.
Permasure: Instant Safe-Wear Times for Chemical Suits.
Assessing real safe-wear times to ensure safe use of chemical suits is not easy. Simply accessing the information required is a challenge.

It works with Lakeland ChemMax 3, 4, Plus and Interceptor Plus ranges and is free to register, download and use.
You can find out more about Permasure® here.
Or find out what you can achieve with Permasure® in our blog here.